–Amin S. Ghabrial, PhD, Assistant Professor of Cell and Developmental Biology
Department: Cell and Developmental Biology
Tubes of different sizes, shapes and cellular architectures compose most organs and glands. How these tubes are formed and what factors regulate their dimensions and their pattern of branching, are among the primary questions that must be addressed before we will understand how organs are made. These questions are also of great medical relevance, because defects in tubes are at the root of many disease states, such as polycystic kidney disease and atherosclerosis, and because the recruitment of capillary tubes to tumors by sprouting angiogenesis plays a pivotal role in cancer. The long-term goal of the lab is to develop an understanding at the molecular level of the basic cell biology of tube formation and branching morphogenesis (the process by which new tubes are induced to bud and branch from pre-existing ones).
We use a simple model tubular organ (the Drosophila tracheal system) to uncover the genetic and molecular basis of how tubes are made and shaped. Using the powerful tools available to a Drosophila geneticist, we are able to manipulate gene function in individual cells and to determine the effects of such manipulations on tube morphogenesis. A large-scale forward genetic screen has been carried out and mutations in roughly 70 genes have been identified that cause striking tracheal defects (manuscript in preparation).
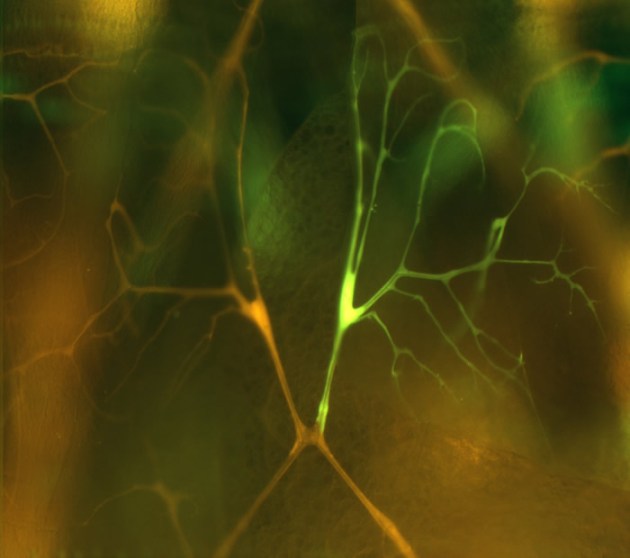
Clones of homozygous cells are generated in heterozygous animals. Homozygous tracheal cells are positively marked with GFP and all tracheal cells are marked with DsRED. Here a single homozygous terminal cell (yellow) is shown. Note that a tube runs the length of each of the fine cellular processes visible in this micrograph. These tubes provide oxygen to the muscles targeted by this terminal cell.
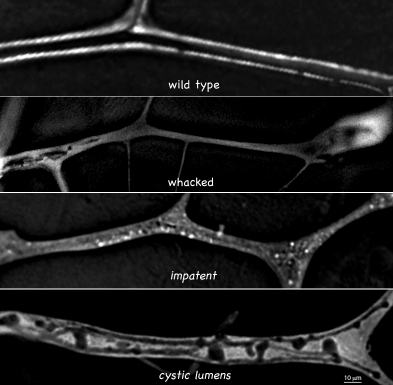
We have focused on the problem of tube formation in tracheal terminal cells (Figure 1). These cells assume stellate shapes, similar to neurons, and build subcellular tubes that extend the length of their dendrite-like cellular processes. These seamless tubes are architecturally similar to the smallest tubes in the vertebrate vascular system. We seek to determine how cells make an internal lumen and in doing so convert cellular extensions into tubes. A related goal is to understand how lumen shape is generated and regulated. As a first step, mutants disrupting these processes (no tracheoles, cystic lumens; Figure 2) will be characterized with the aim of determining the cellular and molecular causes of the mutant phenotypes.
From top to bottom: wild type, whacked, impatent, and cystic lumens tracheal terminal cell branches are shown. Note that the tube lumen in whacked is irregular and comes to a premature dead end, that discontinuous vacuoles fail to coalesce into a tube in impatent, and that the lumen of cystic lumens tubes shows regions of dilation suggestive of abortive side branching.
Another interest in the lab is the process of branching morphogenesis whereby cells bud from the tracheal epithelium and give rise to new tubes. Whereas terminal cell morphogenesis principally involves cells changing shape and topology, formation of larger, multicellular tubes is dependent upon cell rearrangement within the epithelium. We will focus on developing an understanding of how cells change neighbors, and of how they communicate with each other to coordinate their behaviors. Analysis of mutants that are defective in rearrangement (conjoined) and in coordinated behavior (too many leaders) will help guide our dissection of these processes.






Leave a comment