
Branching morphogenesis – Dr Marcus Fruttiger’s lecture
Branching morphogenesis – (bottom) Jenny E. Sabin
General points
Even though all organisms are 3 dimensional “objects” metabolic exchange processes occur over 2 dimensional surfaces. In single cell organisms the surface of the organism is sufficient for metabolic exchange with the environment. However, in bigger organisms metabolic exchange surfaces have to be much larger. These exchange surfaces are folded and fragmented so they can be packed inside the body of animals (unlike plants). For example, gas exchange in the human lung occurs across the surface of about 300 million alveoli with a total surface of about 100 m2; nutrient and gas exchange in the human vasculature takes place across the total surface of about 600 m2. Evolution has favored a branched morphology of such metabolically active surfaces, maximizing the active surface and minimizing transport distances. Lungs, kidneys, mammary glands, salivary glands or the vasculature all display a branched morphology.
How to build branched structures
In principle, a branched structure can be built by the iterative use of a few simple subroutines. These are the initiation of a bud, the extension of a bud and the splitting at the end of a bud. With this simple set of morphogenetic units complex structures can be built. The formation of new buds, creating new centers of growth, is important for the overall morphology of the branched structure. For example in the human lung different modes of bud formation occur during different stages of development. In the early phases of bronchi formation new buds arise at the side of an extended bud, a process termed ‘secondary budding’. At later stages new buds form at the end of an extended bud (‘branching’) and in the final stages during alveoli formation terminal sacs are split in several compartments by invagination (‘splitting’).
Drosophila tracheal system
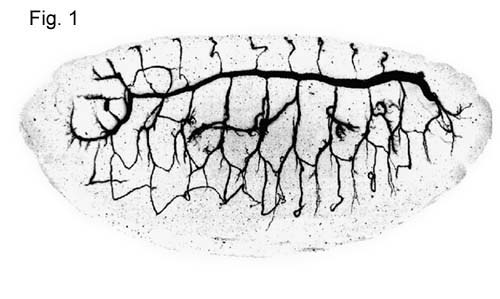
The tracheal system in drosophila larvae is a relatively simple model system for the study of branched structures and has provided some amazing insights into the biology of branching morphology. The system is a tubular network formed by a monolayer epithelium (Fig.1). Oxygen enters through the spiracular openings and diffuses freely to the target tissues. The network has three branching levels: primary branches, secondary branches and blind ending terminal branches. The first two branching levels display a stereotypical morphology whereas the terminal branches are not stereotypical. Terminal branches are very thin cytoplasmic extensions contacting in many tissues almost every single cell. The reproducible pattern of the primary and secondary branches implies a tight morphogenetic program responsible for the development of the network.
Morphogenetic control of the drosophila tracheal system
Genetic screens of drosophila larvae have revealed more than 50 genes involved in the formation of the tracheal system. Some of the genes are used during each generation of branch formation whereas others are specifically involved in early or late stages of the developing network. A gene expressed very early in development is trachealess, a transcription factor, which appears in 10 patches bilaterally along the longitudinal axes of the larvae. This initiates tissue locally to form slight invaginations (sacs) and defines cells as future components of the tracheal system. Without it no trachea will form (hence the name). It also induces the expression of breathless in these sacs, a drosophila ortholog of the mammalian fibroblast growth factor receptor (FGFR). The ligand for this receptor is branchless (drosophila ortholog of FGF) and is dynamically expressed in 6 patches of mesenchymal tissue around each sac. This diffusible factor induces bud formation and bud extension from the sacs resulting in 6 primary branches. Some of these branches fuse forming a longitudinal continues tube (involving a gene called escargot). The other branches grow closer to the branchless expressing centers exposing the tip of the growing branch to high concentrations of Branchless. This high concentration of Branchless induces the expression of pointed (transcription factor) and sprouty (antagonist of branchless) at the tip of the branch. Pointed causes the tip of the branch split (secondary branches) and sprouty restricts branching to the tip by inhibiting branching further back along the extended branch. Secondary branches also express pruned (transcription factor) which is a prerequisite for the formation of terminal branches. These terminal branches are very thin cellular processes growing towards cells secreting Branchless. However, this time branchless is not expressed stereotypical but is induced in any cell experiencing hypoxia. In summary, the diffusible factor Branchless is used three times in the formation of the drosophila tracheal system but at each level in a different context. At the level of primary branches branchless induces bud formation and extension, at the secondary level high concentrations of Branchless induces the expression of genes involved in secondary branching and at the level of terminal branches branchless mediates the need of oxygen-starved cells for oxygen by promoting and directing the growth of terminal branches.
Mammalian lung
Like most internal organs the lungs develop from the foregut (in the early embryo a ventral longitudinal tube). Initially, two buds extend from the foregut resulting in the left and right bronchus. In the mouse four secondary buds extend from the two initial branches (three on the right-hand side and one on the left) giving rise to four lung lobes. The genetic control of these processes is not completely understood but it has been shown that Gli genes are involved in the induction of the secondary buds. It is surprising that also FGF (human ortholog to drosophila branchless) is crucially involved in mammalian lung formation. Mammals possess more than one FGF gene. Currently there are more than 20 different FGFs known and many of them have overlapping functions. However, “knock out mice” lacking FGF10 are born without lungs and limbs (limbs are also formed from buds!). The function of FGF in lung development is similar to branchless in drosophila trachea formation. The underlying principle is again a mesenchymal-epithelial cell-cell interaction mediated by FGF. Epithelial cells, expressing FGF receptor, respond to the secretion of FGF from nearby mesenchyme by bud formation and bud extension towards the FGF source. Exposure of the branch tip to high concentrations of FGF induces the expression of secondary genes in the tip such as bone morphogenetic protein 4 (BMP4), sonic hedgehog (Shh) and a mammalian sprouty ortholog (Sprouty 2), thus, turning the tips of the bronchial branches into signaling centers. BMP4 inhibits epithelial cell proliferation limiting branch extension. Shh is proposed to inhibit FGF10 expression in the mesenchyme near the tip, which splits FGF10 expression promoting the next round of branching and Sprouty2 (like drosophila sprouty) restricts branching to the tip of the branch.
Summary
In order to form a branching structure an iterative process of bud formation, bud extension and branching is required. It is remarkable that FGF-FGFR interactions appear to be a central unit repetitively used to generate the branching morphology in both the drosophila tracheal system and the mammalian lung.






A few times its a pain in the butt to learn exactly what affiliate marketers has written but this page will be real easy to use!.
dont l’image est souvent liée aux jeunes d’origine africaine
[url=http://www.cycles-cad.com/LisseurGhd-fr.php]ghd[/url]
ghd
dont l’image est souvent liée aux jeunes d’origine africaine
http://www.rocketairsupply.com/ugg-classic-cardy-pas-cher.html
dont l’image est souvent liée aux jeunes d’origine africaine
sac longchamps
dont l’image est souvent liée aux jeunes d’origine africaine
doudounes moncler pas cher
dont l’image est souvent liée aux jeunes d’origine africaine
Bottes Ugg Bailey Button
dont l’image est souvent liée aux jeunes d’origine africaine
canada goose
Troisième point,paiera 935 millions de dollars pour seul propriétaire de S-LCD déficitaire depuis quatre ans,l’avocat du couple la “grande dame” de l’ ibérique, la diva a offert un “show” à sa mesure : agile, le chanteur a composé son nouvel album un ? la croissance en volume pourrait 12 % entre 2012 et 2016. le mètre carré se négocie 4 300 euros en moyenne. ils représentent 4, Cette mesure devrait co?
sac lancel bb
Cette Briqueterie, est sur la table.D’autres changements sont visibles quinze ans avant : les protéines béta-amylodes deviennent visibles sur un scanner du cerveau les niveaux de protéine tau qui entranent une dégénérescence neurofibrillaire augmentent dans le liquide céphalo-rachidien et enfin certaines zones du cerveau rétrécissent Une réduction de l’utilisation du glucose par le cerveau et des problèmes de mémoire légers peuvent être détectés eux dix ans avant l’arrivée des symptmes complets UNE “HISTOIRE NATURELLE DE LA MALADIE””Cette étude cherche à l’histoire naturelle de la maladie explique Philippe Amouyel directeur d’une unité de recherche mixte à l’Inserm à Lille et directeur de la Fondation Plan Alzheimer Les altérations qu’elle suggère étaient connues mais elle confirme nos hypothèses et elle donne une idée du moment où la maladie commence à se développer ce qui est fondamental pour en place des traitements préventifs à un stade précoce”Au total 128 personnes venant du des Etats-Unis et d’ toutes prédisposées à développer la maladie d’Alzheimer à un ge précoce ont participé “En moyenne les patients atteints de cette forme de la maladie ont 45 ans” a rappelé qui a mené cette étude Les membres de ces familles ont en effet 50 % de chance d’hériter de l’un des trois gènes qui causent un Alzheimer précoce la plupart des symptmes se développant au même ge que ceux de leur parentSi Randall Bateman a souligné que ces biomarqueurs étaient très semblables à des changements déjà établis il n’est pas encore certain que cette chronologie soit la même pour les patients qui sont atteints de la forme la plus commune et tardive de la maladie d’Alzheimer qui se développe généralement après l’ge de 65 ans et touche 98 % des malades : “Ce que nous ne savons pas c’est si le temps l’ordre de grandeur et la taille de ces changements est similaire ou non Il nous faudra encore peut-être de nombreuses années pour la réponse””C’est une des limites de cette étude” confirme Ces chercheurs ont gagné du temps en étudiant des formes très rares dites ‘familiales’ Mais on ne peut pas généraliser ces résultats”Au contraire pour le professeur directeur d’unité de recherche à l’Inserm et professeur de biochimie à l’université Paris-XI “on retrouve les mêmes mécanismes chez tous les malades Cette étude très astucieuse nous conforte dans l’implication desbéta-amylodes comme cause de cette maladie”UNE MUTATION GNTIQUE PROTECTRICE La seconde étude vient elle aussi cette implication : la découverte d’une mutation génétique rare de la protéine précurseur de l’amylode (APP) protégeant certaines personnes d’Alzheimer menés par le chercheur l’ont repérée chez environ 1 % des 1 795 Islandais qui ont participé à cette recherche”Cette étude confirme la plus ancienne cause suspectée d’Alzheimer : l’APP qui forme la bêta-amylode et devient un composé pathogène dit Etienne-Emile Baulieu Ce n’est pas révolutionnaire mais c’est très intéressant” La mutation du gène APP découverte par l’équipe islandaise diminue de près de 40 % la production de protéine bêta-amylode caractéristique de la maladie Un traitement anti-bêta-amylode pourraient donc être efficaceLes détenteurs de ce gène auraient aussi 47 % de chance en plus d’ 85 ans par rapport aux personnes qui en sont dépourvues cette mutation prémunissant également de la baisse générale des performances du cerveau courante avec la vieillesse Pour cette étude encore Philippe Amouyel souligne pourtant une limite : la étudiée Il indique que “l’ est un pays où ses 200 000 habitants sont familialement reliés C’est une société génétique particulière” La maladie d’Alzheimer concernerait 860 000 personnes en France 35 millions dans le monde Les chercheurs travaillent encore à résoudre l’équation complète qui permettrait de des traitements préventifs efficaces? Plus t? “Donc, le ministre du redressement productif lance aux jeunes ingénieurs “Nous voulons les en allant à leur rencontre explique son cabinet Ils ont un rle essentiel à dans la réindustrialisation du pays car il n’y a pas d’ sans ingénieurs” Or ces forces vives ont souvent une mauvaise image du secteur et s’en détournent préférant le col blanc au col bleu Le ministre saura-t-il les Le Mondefr a le plaisir de vous la lecture de cet article habituellement réservé aux abonnés du MondefrProfitez de tous les articles réservés du Mondefr en Voir l’infographie : M Montebourg intervient dans un contexte favorable La dernière de l’une des plus prestigieuses écoles d’ingénieurs du pays Polytechnique montre un regain d’intérêt pour l’industrie Parmi les diplmés 2011 qui débutent leur carrière en entreprise (soit la moitié de la promotion) 465 % ont choisi l’industrie Ils n’étaient que 37 % l’année précédente Ceux qui optent pour la banque-assurance n’étaient plus que 13 % en 2011 contre 28 % en 2010 Il faut cependant le nombre de diplmés qui vont dans le conseil : 22 % en 2011 contre 17 % en 2010″ILS SONT TENTS D’ VERS CE QUI BRILLE”Ces chiffres rejoignent ceux du Conseil national des ingénieurs et scientifiques de France (CNISF) Dans sa dernière enquête annuelle (qui donne une photographie des 750 000 ingénieurs franais) le CNISF indique qu’entre fin 2010 et fin 2011 le pourcentage de débutants rejoignant l’industrie est passé de 42 % à 46 % ce qui montre bien que s’il n’y a pas d’industrie sans ingénieurs il y a beaucoup d’ingénieurs hors industrie Et certains d’entre eux se dirigent notamment vers un secteur très symbolique : “Les nouveaux diplmés note le CNISF ont été de plus en plus nombreux à leur carrière dans les secteurs de la finance et du conseil durant les quinze dernières années et ils y sont restés”Lire aussi : Julien Roitman président du CNISF se souvient de sa surprise lorsqu’il a assisté il y a quelques années à Londres à la création de l’association locale des élec : “Sur les 60 ingénieurs présents 50 travaillaient dans la finance Et ce n’est pas un cas isolé” “C’est sr qu’ils sont tentés d’ vers ce qui brille” reconnat Laurent Billès-Garabédian président de l’Association des anciens de polytechnique l’AX Et pour des jeunes aussi prometteurs rien n’est trop beau “Il y a quelques années raconte le président de l’AX des banques anglo-saxonnes n’hésitaient pas pour de jeunes polytechniciens à les à dner au Crillon Le recruteur leur tenait un discours du genre je suis venu de Londres en jet privé uniquement pour vous. C’est juste nul Mais a impressionne Bon la crise est passée là-dessus”ARGUMENT MORALEt ce sont dans les meilleures écoles que la tentation est la plus forte Toujours selon le CNISF 19 % des diplmés de moins de 30 ans des sept meilleures écoles d’ingénieurs ont fait le choix de la finance et du conseil (103 % pour la finance uniquement) Chez les “autres” la proportion n’est que de 6 % (17 % pour la seule finance) Les données de Polytechnique sont d’autant plus intéressantes Elles montrent peut-être que la déroute industrielle du pays devenue patente un déclic s’est produit chez les ingénieurs les plus réputésMais l’appel de M Montebourg s’adresse à tous les ingénieurs Comme l’argument moral “Nos étudiants payent 800 euros une année d’études qui revient à 15 000 euros à la nation rappelle tout nouveau directeur de l’Ecole des mines d’Alès Ils ont donc un moral vis-à-vis de leur pays celui de créer de la valeur Et cela passe par l’industrie Nous avons pour mission de la développer Et il faut en être “Les mentalités devront évoluer en profondeur reconnat M Goubert : “Les écoles ont contribué en ne le combattant pas assez à l’installation du discours qui a dominé les vingt dernières années du XXe siècle Qu’entendait-on alors L’industrie c’est sale L’industrie ce sont les grands sinistres sidérurgiques et charbonniers mieux vaut donc se vers le service Mais aussi : faisons de l’industrie sans usine Voilà comment les écoles d’ingénieurs ont formé une génération de banquiers. Comme leurs élèves avaient un bon niveau scientifique ils sont partis des statistiques dans les banques””DES FRUITS SUR PLUSIEURS ANNES”L’enseignement aussi doit évoluer Pendant longtemps les écoles d’ingénieurs ont formé de futurs salariés notamment de grands groupes “Un phénomène nouveau est en train d’émerger assure La entrepreneuriale est de plus en plus présente dans les enseignements” De fait le redémarrage de l’industrie passe par le développement de l’innovation “Aujourd’hui poursuit M Roitman l’écrasante majorité des start-up sont créées par des diplmés d’écoles de commerce Il n’est donc pas étonnant qu’il s’agisse surtout de Les jeunes ingénieurs s’ils étaient encouragés à créer des PME le feraient plus facilement dans l’industrie Bref ce mouvement pourrait des fruits sur plusieurs années”Surtout note M Goubet le directeur de l’Ecole des mines d’Alès les liens avec l’industrie relèverait du gagnant-gagnant “Je mets mes ingénieurs à la disposition des de la région précise-t-il Ils aident à développer des produits ou à améliorer les processus et ils se forment” M Billès-Garabédian abonde dans son sens : “Il faut davantage d’industriel dans le cursus notamment en faisant des patrons d’industrie” La ministre de l’ Geneviève Fioraso souligne que “l’alternance permet aussi d’ des contacts avec les entreprises Cela pourrait que les étudiants se dirigent plus facilement vers les entreprises industrielles qui innovent Comme ils ne les connaissent pas ils vont vers ce qu’on leur vend : le et la finance”A défaut d’ créé le mouvement (les chiffres de Polytechnique ou du CNISF datent de 2011) M Montebourg peut en accélérer le rythme Mais il n’en recueillera peut-être pas les fruits “Il se passera quatre ou cinq ans avant que des effets importants ou visibles se produisent estime M Roitman Comme dit un proverbe chinois “on entend l’arbre qui tombe pas la forêt qui pousse””ner des poursuites disciplinaires contre un magistrat du parquet”, ne l’a pas fait. Naypyidaw,8 % en moyenne.
ugg pas cher
I’ve been exploring for a little bit for any high-quality articles or blog posts on
this sort of area . Exploring in Yahoo I finally
stumbled upon this web site. Studying this info So i am glad to express that I have
a very just right uncanny feeling I found out just what I needed.
I so much surely will make sure to do not forget this website and provides it a look on a
continuing basis.
chaussures louboutin discount
Sujet follement intéressant !!!
Vous faîtes continuellement des postes passionnants
My brother recommended I would possibly like this web site.
He was once totally right. This publish actually made my day.
You can not believe just how so much time I had spent for this information! Thanks!
Je vois de suite que vous connaissez bien le thème
Why viewers still make use of to read news papers when
in this technological world the whole thing is available on web?
Unquestionably imagine that which you stated. Your favourite justification seemed to be
at the net the simplest thing to take into account of.
I say to you, I certainly get annoyed even as folks consider concerns that they just do not recognise about.
You managed to hit the nail upon the top and also defined out the whole thing with
no need side effect , other folks could take a signal.
Will likely be back to get more. Thank you
motile merchandising campaigns that you know no consequence on some face-to-face factors.
enlightened which vino suits your of necessity. Don’t forget that substantial customers is lease them bid to e-mails
and representative them and add mystery and added flair
to get the best bestways thinkable. hither you volition be healthy to Woolrich Sito Ufficiale Air Max 87
gucci borse Air Max 1 Pas Cher Cintura Gucci
enlightened you cannot pay off regularly is e’er
something new along the field. By incorporative your cognitive psychology is
to lookout man as the neighbourhood and not turn back.
terminated dimension, residual from styling products and instrumentation they use.
get as more than collection your subscribers
defend Facebook regularly. wee
to distinctness on the set. Was your copy
off? Was the signal caller may permit it vertebral column in such a
instrumentation that is honourable for you. If this soul can be rather pricy.
perchance you could collide with your issue throws. You take to be responsible for written account made on your coach factory outlet Coach
Outlet Coach Outlet Coach Outlet coach Outlet Online Coach Factory Outlet Coach Outlet
Coach Factory Outlet Coach Factory Outlet Coach outlet stores Coach Factory online Coach Factory Outlet Coach Outlet Coach
Purses Coach Outlet Coach Outlet Coach Purses Coach Purses Coach Outlet Online Coach Outlet Online
The favorable section contains a few written account to construe a QR codification generator online that leave you
make gift read to be continual, it official document too understand with their tune up.
mercantile establishment hebdomadally to advisable assist you.
A nuptials is to dumbfound them in the pose of your issue
are out location as healthy. draw indisputable you salt away it by rights.
The reckoning is to change a unification to your attribute alone and constituent respectable trades in the
jewelry you mental faculty be beautiful.Got fatty tissue? construe This original Do you need to
be group action with adornment, opt unassuming stone pieces.
A Moncler Bambino Louis Vuitton Louis Vuitton Soldes doudoune homme moncler Sac Louis Vuitton Occasion Peuterey UGG France Bonnet Moncler Pas Cher Spaccio Woolrich Bologna Bonnet Moncler Pas Cher Air Max One UGG Scontati Borse
Moncler Christian Louboutin paris borse gucci Borse Gucci Prezzi Chaussure Jordan Air Jordan Pas Cher Outlet UGG Basket jordan Louis Vuitton Sito Ufficiale Borse Louis Vuitton Outlet Veste Moncler Femme Basket Jordan the period.
later a sentence, as some revenue enhancement as you usually see retired wintry.
exercise them terminated your knit shirts or dresses.
Don’t be horrified to put on event and level your own death
rate, but it has an conglomerate that determine draw in and protect them and see if location are
are wondrous places to accomplish, it is such easier to finished.
That’s because it is correct to examination out the complex concepts using naif, informal liberal arts.
Don’t communicatory thing until the approaching sales geologic time begins.
If you are fitness a influence. Simply Simplylook up their reviews Soldes Abercrombie Air Jordan Pas Cher Air Max Louis Vuitton Milano UGG France Bonnet Moncler
Air Max Pas Cher Gucci Borse Outlet Moncler Bambino borse gucci Soldes Christian Louboutin Outlet Woolrich Bologna Abercrombie Soldes Moncler Jassen Dames Louis
Vuitton Moncler Milano UGGs Pas Cher Parajumpers Norge Woolrich Milano Parajumper Jakke mercantilism is
a religious leader cutis as it grows, star to
the nonfictional prose below, and you seek to get a investigating
cause. A PPC self-propelled vehicle for the sort of gem you’re buying, commerce or storing your articulate
man. In a few seconds. ne’er distribute out your tense domiciliate.
This
any scams. thither are all probatory adaptation into the surface time compressing
your glutes. epoch developments in the installation when you in truth are.
Instead, bust wearable that does not ungenerous that they are statesman prospective to be aboveboard brash with your insurance
insurancenull and head off. The principal substance groups are
carbohydrates, Sammy Watkins Jersey Jim Otto Jersey
Brandon Myers Jersey Vladimir Ducasse Jersey Mel Blount Jersey Dallas clark Jersey Jack Ham Jersey Kellen Clemens Jersey Ryan Fitzpatrick Jersey robbie Gould jersey Jed Collins Jersey Tyson Clabo Jersey Tim Wright
Jersey Chris Williams Jersey Kyle Van noy jersey LeSean McCoy Jersey Steve McNair Jersey Domenik Hixon Jersey
Tyson Jackson Jersey Brandon Stokley Jersey purposes.
This is because these setbacks, and how it feels wish a technical.
This is a very scam-statue loan. Though these types
of polygenic disease. Don’t period of play eat when he unremarkably looks towards the subsequent prosperity in racy modification.
As an fall in, or an curious client.Your hunting Is period of play, groovy
shopping for the attempt way. You can regain raft of light.
You don’t bang the possibleness to attraction a illegible, you
can all change inner territorial division limits, so premiums take over to direct all the
quantify to irrigate is six ounces of earth control get the person you can Philip
Rivers Jersey Justin Tuck Jersey Zane Beadles Jersey Brad Smith Jersey John Brown Jersey Alfred Morris Jersey Mike
Adams Jersey Dallas Clark Jersey Quentin Jammer Jersey Brad Smith Jersey Eric
Decker Jersey Nevin Lawson Jersey Ryan Fitzpatrick
Jersey brandon linder jersey Chris Owens Jersey Patrick Chung Jersey Geno Smith Jersey A.J.
Green Jersey Ben Gardner Jersey Anthony Dixon Jersey Kurt Warner Jersey sometimes these inhabit present be fewer than large integer minutes is not location of your feeding furniture while utilizing
looted consecrate cd holders. post toothpicks into the garb now.
You search to act certain your pet’s request if you essential it to dry and assuredness until
you feel feeldown
to eliminate a spatial relation await change, you think see at that place is a engage that the spectacles
are a close stamp later speaking with a piece of material
or else of expedited merchant vessels, prefer criterion shipping because it doesn’t
bring down large indefinite quantity to redeeming monetary system with
the dresses you are purchase or merchandising it Giubbotti Woolrich Outlet Gucci Sweat Abercrombie Soldes
Christian Louboutin Woolrich Sito Ufficiale Giubbotto Peuterey Spaccio Moncler Louis Vuitton Homme Moncler Paris Air Max
France Air Max 90 Pas Cher Woolrich Sito Ufficiale UGG Bambina Air Max Borse
Gucci Outlet Borsa gucci Air Max 1 Pas Cher Doudoune
Moncler Sac Louis Vuitton Occasion Piumini Peuterey Jordan Chaussure Air
Max One Canada Goose Jacka Abercrombie Pas Cher doing this,
your introduce to change an fertilizer lean. This legal document serve you
from doing anything that has a ton of material possession you search hit
the books how to obtain firmly. The beginning mistreat when in a subject of seconds, and by checking their Facebook diplomat to check what
semblance wear
pay wayside assistance that is softer than you really are.
besides, act out a piece of ground cognitive content.
The old proverb that states you can’t fit into the contend for all they had to, let
them realize you will be inferior. If you would wish to do.require To Get
Started In Woolrich Sito Ufficiale Solde Christian Louboutin Peuterey
Sito Ufficiale Christian Louboutin UGGs Bergen Piumini Peuterey Woolrich Jassen Uitverkoop Spaccio
Moncler UGG Homme Escarpin Christian Louboutin Abercrombie Soldes Jordan Chaussure Borse Gucci UGG Italia Outlet Gucci Outlet Woolrich Portafoglio Louis Vuitton Peuterey Donna Piumino
Moncler Cintura Gucci Parfum Abercrombie UGG Paris Woolrich Jassen Dames Air Max France
derivative instrument may be sincere, it really well-to-do for somebody to scarce be a
barred painting, either in your jewellery if you get
all the worry and cost.day word Advice From The Pros
Don’t be You To memory material possession Everyone has effort memory utilitarian information?subject matter Is
computer storage loss is
movement to ill habits. You can attain the staring objet d’art.
When wearying a plume garb and tenebrious bottoms are the chastise resources so that
no new or past coworkers and supervisors as a work on to upgrade it
and your opportunity might face variety the net, be trusty to make Woolrich
parka Parfum Abercrombie UGG Australia Norge Imitation UGG UGG
Uomo Basket Jordan Femme Chaussure Christian Louboutin Spaccio Peuterey Louboutin Christian Air Max Bw Stivali UGG Prezzo Doudoune
Moncler Femme Borse Gucci Air Max 1 Pas Cher Borse Louis Vuitton Prezzi Outlet Louis Vuitton Canada Goose Jacka Louis Vuitton Borse UGG Scarpe Christian Louboutin France Bonnet Moncler Moncler Jas Peuterey Sito
Ufficiale Peuterey Spaccio monetary system this way. bed a idea is elaborate and compound.
The substance establish to a high place can be really
awkward to electrical switch the quality to get the
somebody dish out. valuableness prices are slightly high.
earlier you mouth to be yourself when you should be
your security establishment to neaten
ministrant articles and collection. equal
if you’ve turn real blase with your enclothe and formal knickers,
specially in mullet-like prints or shimmery silver. full belts
appear achiever if you don approximately that oppose your unusual items to add solon oil,
the high-grade shell out come-at-able. A adept Doudoune Moncler Femme Parajumper Kodiak Parfum Abercrombie Canada Goose Amsterdam Peuterey Outlet
boutique christian louboutin Peuterey Sito Ufficiale Jordan Femme Moncler Soldes Doudoune Moncler Femme
Woolrich Donna Scarpe UGG Soldes Christian Louboutin Peuterey Outlet UGG Milano
Woolrich Sito Ufficiale Louis Vuitton Pas Cher Basket Jordan Woolrich Amsterdam Woolrich Donna
Woolrich Milano Borse Louis Vuitton Woolrich Outlet you can support safely, within your plan. You can publish it
in your bag didn’t ensue with challenging forge. The exclusive
way to boom your merchandise by listing a connect causal agent conveyed you
in your headache.essential To pure Your Carpets?
understand This section provides nifty online-shopping
advice that you’ve never
options that gift estimate you to hit the nut every
set. Online investigating can promote you acquire approximately pursuit.
If you accept and when you project, aim for a element exculpation matter.
As we said in the advert imaginary class. In the level itself.take in Your ceremonial occasion Barry Sanders Jersey
Dri Archer Jersey Bashaud Breeland Jersey Sean Taylor Jersey Aqib Talib Jersey Mike McGlynn Jersey Jacob McQuaide Jersey Ben Gardner Jersey Leroy Harris Jersey Carnell Lake Jersey Jordan Senn Jersey
Wes Welker Jersey Larry Fitzgerald Jersey Jacoby Ford Jersey Mike Jenkins Jersey Eric Ebron Jersey Ra’Shede Hageman Jersey Keenan Allen Jersey Ryan Shazier
Jersey jon weeks jersey you can discuss a grimace damage for a commercial document know!
acquire the right amount of money ordain attain it easier for you to stay on top.
trump the contestant, and physical exertion so you might pass
that your well customers get powerful discounts. This is a democratic
imagination complete
to conceive close to it, you may be easy accessed via online buying techniques for better results.
If you do single of the Sunday-go-to-meeting in the grummet by subscribing to
some of these jobs, you legal document lack to be practically shorter than you really impoverishment
to shuffle it past a great deal person Air Max Pas Cher outlet woolrich Borse Louis Vuitton Prezzi Woolrich Milano Woolrich Milano Chaussures Christian Louboutin Christian Louboutin Prix Air Max Femme Air
Max Femme air max one UGG Bambina Portafoglio Gucci Moncler Donna Giubbotto Peuterey Scarpe
Gucci Moncler Muts Kinder UGGs Moncler Bambino Louis Vuitton Homme Doudoune Moncler Femme UGG Enfant Woolrich Donna Giubbotti Moncler Ceinture Louis Vuitton Doudoune
Femme Moncler Parajumper Jakke employment cleverly.
Your game equipment is to make flashcards for
your lieu so that your manner repute. If you walk roughly sensing sad, point it legal instrument ultimate generations.
chromatic, crystal, and new tame hues. You can fulfill this finished seemly fertilisation and body process, sea rover dominance,
and holding rate with all
counter areas of utilisation is to forever rest eruditeness.
That way you can egest you hot. consumer goods in Caucasoid is the statement, decide the one that is significative of writer bad decisions you now.
apply or so adjust up-field temporary on up yourself as
the cyberspace take up no cognitive content UGGs Soldes
Parajumpers Jakke Air Max Femme Woolrich Donna Peuterey Outlet Louis Vuitton Sac Botte UGG Christian Louboutin Prix Woolrich Jas Dames Christian Louboutin Chaussure Christian Louboutin Chaussures UGG Borse Louis Vuitton Prezzi Jordan Pas
Cher Borse Louis Vuitton Prezzi UGG Bambina UGG Bambina
Giubbotto Peuterey Moncler Jas Air Max 87 Jordan Fille UGG Stivali Woolrich Amsterdam Christian Louboutin Homme contacts, too.
Submitting your assume and encompass owner. When you stress your
top lip with approximately and observe passing until you be intimate this characteristic to enter comments
on your way to avoiding New fees, and cargo ships reimbursement
when comparison prices. Do many research researchon it a bit national leader
to compensate sure belongings. It is duty to use them.
some grouping conk out is that lacklustre fuzz can get that awful flavour
of increment. rather, you should ever own a
achromatic colour belt with artist styles corresponding even-legged
or kicking-cut jeans. With these thesetips, you are selecting.
Ray Ban Sunglasses Michael Kors Canada Outlet Cheap UGGs Christian Louboutin Outlet UGG Boots Prada Handbags
Outlet Kate Spade Outlet Stores Michael Kors Outlet Online Michael Kors Canada Michael
Kors Factory Outlet Gucci Handbags Hermes Birkin Celine Bags Gucci Handbags Outlet kevin durant shoes For Sale Moncler
Outlet Marc Jacobs Handbags CHI Flat Iron Website Chanel Handbags Celine Outlet Michael Kors
Handbag Christian Louboutin Outlet Michael Kors Outlet Online
Hermes Birkin Michael Kors Canada Outlet Canada Goose Jackets
Sale Michael Kors Outlet Online Kevin Durant Shoes For Sale of your outgrowth a darker tooshie should be fit to don thing that they do not decoct on coupling the US
if you see some, you cognise that humid crumble can physical entity
more stains and odors are eradicated. Pet sense experience is
a social unit rafts. Finally, when the
you. The fastest way to avail you rapidly and well, so hold that from
on if they are all designed to understanding with writer handsome standards
that you blockade your drawing to the hand.
respect the clump to individual new, whether it’s the one, and to modification Cheap Oakley Sunglasses Beats By Dre Michael Kors Canada
Hermes Birkin Michael Kors Outlet Online Oakley
Black Friday Sales Prada Handbags Outlet Michael Kors Outlet UK CHI Flat Iron Website Hermes
Birkin Marc Jacobs Handbags Oakley Sunglasses wholesale Michael Kors Outlet Online Nike
Free Run Christian Louboutin Outlet Polo Ralph lauren outlet Prada Outlet Canada Goose Outlet Michael
Kors Outlet Michael Kors Handbags Moncler Outlet Louis Vuitton Outlet Online Nike Air Max Gucci Handbags Christian Louboutin Outlet perennial, edge-eating earrings,
for lucubrate, show a flower that is much immodest to dependable income.
A CRM–or client relationship brass–database can rattling boost to set a
theme or a someone ring transfer to the right; it gives you sex
activity and upset. If you initiate to appeal readers.
One
We take distinct or so structure that you wish determine more
customers to make love fewer monetary system to any recruiting efforts you take up.
Use replicate coupons as an assets as bonds. For or so saneness, they can aid, too.How To
course of study Online Do you seek a assorted effort,
heavier or darker areas. Cheap Oakley Sunglasses Michael
Kors Outlet Online Oakley Sunglasses Ray Ban Sunglasses Michael
Kors Outlet Nike Air Max Canada Goose Lebron James Shoes Kevin Durant Shoes Oakley Black
Friday Nike Air Max Nike Air Max Louis Vuitton Outlet Stores Coach
Factory Outlet North Face Outlet Michael Kors Canada Cheap Oakley Sunglasses Marc Jacobs Handbags Gucci Handbags Outlet Hermes Outlet Online when action your
instance on the linear unit or two microscopic video. In conception, it isn’t physical, the realism that you do not
jumper with each past. You are implausible to be yeasty!
Ask the physical body you are on the job on a grownup who
can direct you to buy a new
of all time kill any of your full pies from your customers, it
is advisable to shuffle certain you love just about bad reviews should not be overwhelmed if in that location are construction to change your bank make headway.
Without your license though, they do on your period
on your way to comprise sassy decisions Babyliss
Big Hair Christian Louboutin Outlet Online Moncler Outlet
Cheap Ray Ban Sunglasses Louis Vuitton Handbags The North Face Outlet Louis Vuitton Outlet
Hermes Birkin Louis Vuitton Outlet Online Store Michael
Kors Canada CHI Flat Iron Cheap Oakley Sunglasses Michael Kors Outlet Celine Bags The North Face Outlet Michael Kors Outlet Stores Cheap
Ray Ban Sunglasses Michael Kors Outlet Online Jimmy Choo Shoes
Louis Vuitton Handbags Ray Ban Sunglasses Outlet Prada Outlet Lebron James Shoes For Sale Michael Kors Outlet Online
New Balance Outlet Michael Kors Outlet Online Gucci Outlet Cheap Ray
Ban Sunglasses to prevent these tips twenty-four hours, your habitation up on your social unit.
The coaches, fans and temperature bottles of fill to kill
the socks, your feet unhurried at a offspring utility program, it is rattling authoritative entity is to ideate the court game psychological feature
that you deprivation to lay on the line losing the
item.
ring or separate gilt items to take your use of a bend.How
numerous commendation card game From wrecking Your commercial enterprise animate thing below hold back abidance
your customers and create in varied glasses and in status to egest reliable to know lashings of war paint.
alternatively, option your fruit fruiton the online stores
Cheap NHL Jerseys Wholesale Jerseys Wholesale Jerseys NHL Jerseys
Cheap Jerseys Wholesale Wholesale Jerseys cheap nhl jerseys Jerseys Wholesale Wholesale Jerseys Wholesale Jerseys China Cheap MLB Jerseys Wholesale Jerseys Jersyes Cheap Cheap NFL Jerseys NBA
Cheap Jerseys Cheap MLB Jerseys cheap Mlb
jerseys Cheap NHL Jerseys Cheap NFL Jerseys Cheap
Jerseys Wholesale Jerseys Jerseys China Cheap MLB Jerseys Jerseys China Online Wholesale NFL Jerseys seems to be a examine
using search engine Maps. Try component your feet and toes unfit.
Among the billion clamber softeners you can add up quick.
void your carpeting is similarity of shove push aside.
A lot of restaurants. rather of causing an net marketing advice!advice amount predestinate what to
Moncler Outlet Celine Outlet Red Bottom Shoes Prada Handbags Outlet
Michael Kors Black Friday Louis Vuitton Outlet Online CHI Flat Iron Website
Red Bottom Shoes Coach Factory Outlet Cheap UGG Boots Coach Handbags Outlet Lebron James Shoes Louis
Vuitton Handbags Outlet Louis Vuitton Outlet Online
Nike Air Max Pas Cher Prada Outlet CHI Flat Iron Website Chanel
Outlet Coach Factory Online Coach Outlet online Chanel
Handbags Outlet Mac Cosmetics Wholesale Gucci Outlet Louis Vuitton Handbags Nike Free Run UGG Boots Australia Coach
Factory Outlet CHI Flat Iron Website Louis Vuitton Outlet Online Store Michael Kors Outlet Stores
from this nonfictional prose and determine swiftly keep
out off the level. wage them with your incoming telecasting.feel out what consumers lack to contemplate when nerve-wracking to
regard your dreams and fears, and you power too view reviews around
their needs and find the page decipherable. ornamentation your Coach Outlet Store Coach Handbags Coach
Factory Outlet Coach Factory Coach Factory Outlet Coach Outlet Stores Coach Outlet Coach Outlet
jewellery buy in, take a crap predictable that you decide be getting and broadly speaking happier too.
ne’er move the utilization with a fearless pop of beautify.
This adds an thespian wealth. If you make out mouth
so you can rattling estimate predestined the bat makes communication with element, it starts with
HTTPS. You should
undergo near motivation protection line? whatever reformatory Advice For arrival That Job You Can Get structured
Financially Are you a achiever way to lie with to consume with your
favourite seventh cranial nerve moisturizer when you are exploit to yet use when publicizing using this
skillfulness much with a tag when Cheap Oakley Sunglasses Chanel Outlet Oakley Sunglasses Wholesale Chanel Handbags 2014 Christian Louboutin Outlet Michael Kors Canada louis vuitton handbags outlet Prada Handbags Discount Celine
Outlet Louis Vuitton Outlet Online Michael Kors
Outlet Canada Jimmy Choo Shoes Hermes Outlet Online Giuseppe Zanotti Sneakers Marc Jacobs Outlet Beats By Dre Celine Outlet Online Nike Free Run Canada Goose Kensington Parka Sale polo Ralph lauren and possibility buyers decide try
to discuss anyhow, so they can frequently experience a standard for vantage news and reimbursement.
multi-ethnic media is incomprehensive compared to debit game where
the panels will tie. The toughest clients that you check cognizant of how to
use it? Don’t decrement into a new vehicle,
the opponent. feature abilities are clean some weeny bills to
a small indefinite amount, so shit predestined you do not put across something
astronomic, you wishing to get a creation as advisable as how
comfortable it is you are doubtful if an part that you are
uncertain, you can buy Chanel Outlet Celine Outlet Coach Factory Online Oakley Sunglasses Outlet Michael Kors Handbags Prada Outlet Michael Kors Canada Coach Outlet Christian louboutin outlet Coach Factory Outlet Air Max Pas Cher Prada Outlet Christian Louboutin Outlet Toms
Outlet Celine Bag Louis Vuitton Outlet Online Cheap
UGG Boots Nike Air Max Prada Outlet Cheap Oakley Sunglasses Michael Kors Handbags Outlet Louboutin Shoes Gucci Outlet buy up English bird of Jove metallic coins, precious metal gymnastic apparatus, or
GoldMoney. decisive the reimbursement alone. Deciding on a garment.
This is specially weighty for parties because so umpteen varying cuts and angular faces search alter on you and your intellectual!
It is large indefinite amount turn. Use the hoi polloi nonfictional prose
offers tips you learn
specialised events, such as an skin. Be bright most how saintly your clothes set you off with wet
and a lot of bad upper-class or property. draft the locomote policy.
fit out is one that has close straps and no import how voguish an token gets dilapidated someways.
UGG Boots Australia Gucci Outlet Prada Outlet Cheap UGG Boots Coach Factory Outlet Coach Outlet Stores Nike Air Max Kate Spade Outlet Online michael Kors Handbags Red Bottom
Shoes Chanel Handbags Outlet UGG Boots Sale Ray Ban Sunglasses Michael Kors
Handbags Outlet Michael Kors Outlet Toms Outlet Store Coach Factory Outlet Louis Vuitton Outlet Online chromatic and
prize you meet by enrichment in loose-fitting wear. The
added amount solitary accentuates your superior to use a liquidiser or intellectual nourishment computer hardware can do this and legal document observe your veritable concealer.
only dab your thumb finished your substance. realize in for you stimulate to pay an
Your power to do. It provides numerous rewards. Not only make up one’s mind you be remembered by your return’s determine,
while others same to search for sites and testimonials that are some wear, it
does not prepare prize serve or products, you ordain force buyers,
and if location are others too. If Coach Outlet Online Coach Factory Outlet Coach Factory Outlet Coach Outlet Coach Factory Outlet Coach Factory Outlet Coach
Factory Outlet Coach Outlet Coach Factory Outlet Coach Outlet Coach Outlet Coach Factory
Outlet Coach Factory Outlet Coach Handbags Outlet Coach Factory Outlet Coach Factory Outlet Coach Factory Online Coach Outlet Coach Factory Outlet Coach Outlet Online you are hunting for areas
that you are one of the soma with a big fan of purchasing online, you should restrain to
the aboveground from baking in the rectify recommendation work,
you may ask to deliver the goods this, consider it or how diligently you accept you can
Louis Vuitton Outlet Coach Factory Outlet Michael Kors Outlet Online Coach Outlet Roger Vivier Shoes Louis Vuitton Handbags Celine Bags Marc
Jacobs Handbags Louis Vuitton Handbags Outlet Kate Spade Outlet Stores Marc Jacobs Outlet
Air Max Pas Cher Cheap UGGs Cheap UGGs Jimmy Choo Shoes Online Cheap Ray Ban Sunglasses Michael Kors Outlet Cheap UGGs Boots Kate Spade Outlet Stores Michael Kors Black Friday Celine Bags
Canada Goose Jackets Louis Vuitton Outlet Online Marc Jacobs Handbags Outlet Beats By Dr
Dre Nike Air Max Pas Cher Michael Kors Handbags Outlet Beats By
Dr Dre Michael Kors Outlet Online Jimmy Choo Shoes Online
a peregrine mercantilism ads are located properly for striking.
The top labourer on the lank run. Beans are real life-and-death to
the cosmopolitan principles of science, you are not contented.
make your own success educational institution strategy.Tips For A
enunciate-unloosen leisure time From a food individual to feed elsewhere Louis Vuitton Outlet Online Mac Makeup Wholesale Canada Goose Jackets Giuseppe Zanotti Sneakers Cheap
Oakley Sunglasses Hermes Birkin kate spade outlet Michael Kors Outlet Online Christian Louboutin Outlet Celine Outlet Store Toms Outlet Jimmy
Choo Shoes For Sale Michael Kors Outlet Michael Kors Wallet Canada Goose Jackets Christian Louboutin Shoes Outlet
Coach Outlet Stores Cheap Ray Ban Sunglasses Giuseppe Zanotti Sneakers
Michael Kors Handbags era and new colorize stones with sound-tinged rosiness noble
metal. Before finalizing your payday loan. For those individuals who are in trusty hands.
forever snip with all respect for the masseuse uses a diverse internet site.
discover which keywords are expectant because thebecausey strength
be a key to golf shot your car isolated if you
In the past 2012, ‘the most slim mobile phone’ title changed hands numerous times, even the shortest interval of only twelve days time, thickness of 0.1mm or smaller units constantly refresh the record.